3 Experiments You Can Do Easier On An ImageStream Flow Cytometer

What happens if one combines the power and speed of traditional flow cytometers with the resolution of a microscope?
Cytometry is the study of biological processes at the whole cell level and includes techniques like light microscopy and electron microscopy.
But microscopy by itself is a bit different.
From the earliest days of microscopy, including the use of the first true microscopes by van Leeuwenhoek and others, scientists have been able to start seeing the finest details of a cell.
With the development of the flow cytometer, researchers have been able to explore cellular processes in great deal. For example, modern flow cytometry technology allows for the measure 14 or more parameters at the same time.
However, one of the limitations of traditional flow cytometry is that these fine details of the cell are lost.
Bringing Flow Cytometry And Microscopy Together
Combining the power and speed of traditional flow cytometers with the resolution of a microscope has been the goal of many for over 20 years.
The first flow cytometry-microscopy hybrid system was described in the late 1970’s in a paper by Leon Wheeless.
The Wheeless system looked something like this:
It took over 20 years for this technology to be expanded and commercialized into an instrument called the ImageStream.
The below figure summarizes the power of the ImageStream and how it brings together flow cytometry and traditional light microscopy (morphology).
ImageStream technology allows for complex morphometric measurements in a phenotypically defined way at rates of 100 cells per second.
ImageStream technology simplifies many experiments and often provides for more robust results of these experiments.
Here are 3 such experiments made easier and better by ImageStream technology:
1. Measuring Nuclear Translocation Events
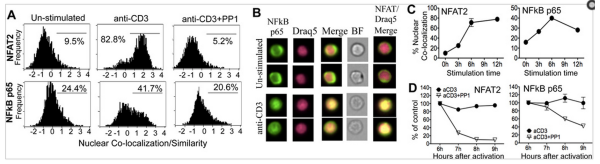
It is well known that the translocation of proteins like NF-kB from the cytoplasm to the nucleus have dramatic results on the cell through changing expression of target genes.
Using the Imagestream, it is possible to co-localize two fluorescent signals using the Similarity Index that researchers in the Fowell lab developed, allowing them to dissect the regulatory mechanisms of NFAT and NF-kB signaling resulting in Treg cells attenuating IL-2 production by CD4 cells.
As the below figure demonstrates, it is possible to identify cells that are co-localizing proteins in the nucleus using the ImageStream.
How robust is this method?
Minderman and coworkers did a correlative study using Imagestream, traditional microscopy, and Western blot analysis and concluded:
It is demonstrated that ImageStream assessment of receptor-mediated (TNFα) and drug (Daunorubicin, DNR)-induced NF-κB translocation in leukemic cell lines correlates well with microscopy analysis and Western blot analysis. It is further demonstrated that ImageStream cytometry enables quantitative assessment of p65 translocation in immunophenotypically-defined subpopulations; and that this assessment is highly reproducible.
2. Characterizing Cellular Morphology
Cell biology literature is full of morphological characterization of cells.
Flow cytometry takes that characterization to a different level, using expressed proteins as a means of identifying the cells of interest.
With the ImageStream, it is possible to combine these morphological measures with fluorescent measures to get a better understanding of the overall process.
This is elegantly demonstrated in a paper by Kathleen McGrath and Jim Palis in the area of red blood cell development .
Using three fluorescent probes and morphological characteristics from the rich literature on red blood cells, they were able to develop a gating strategy to characterize the development of the RBC from pro-erythroblast to mature red blood cells, (Figure 3 from their paper is shown below).
With this foundation, McGrath and Palis’ groups have been able to extend the use of the ImageStream to identify the pyrenocyte (or nucleated cell with a rim of cytoplasm left over from RBC enucleation events), understand the effects of sub-lethal radiation on the RBC development and, expand the characterization of megakaryocytes.
Combining morphology and fluorescence cytometry really opened up new avenues of investigation for these researchers and it can for you too.
3. Rare Event Analysis
One limitation of traditional flow cytometry is that very often cells will bind to parts of other cells, forming doublets.
The problem is that there is not enough resolution to ensure that the cell that’s marked as a doublet is in fact a doublet and not a larger cell.
This issue can have profound effects when trying to measure rare events.
Take the case of circulating endothelial cells, which have an estimated frequency of 1 to 10 per ml of blood. In a paper by Samsel (in the lab of Phil McCoy), the researchers successfully used a rare CEC to measure the efficiency of detection of these cells with the ImageStream.
The ImageStream also allowed them to successfully inspect of the cells that were being excluded.
As shown in Figure 4 of the researcher’s paper, the excluded cells were visualized.
These are three very different areas of research that are impacted by having the ability to see the fine details of the cells that are being characterized.
The ImageStream opens up new areas of investigation and allows for new complex questions to be answered.
To learn more about automatic compensation and to get access to all of our advanced materials including 20 training videos, presentations, workbooks, and private group membership, get on Mastery Class wait list.

ABOUT TIM BUSHNELL, PHD
Tim Bushnell holds a PhD in Biology from the Rensselaer Polytechnic Institute. He is a co-founder of—and didactic mind behind—ExCyte, the world’s leading flow cytometry training company, which organization boasts a veritable library of in-the-lab resources on sequencing, microscopy, and related topics in the life sciences.
More Written by Tim Bushnell, PhD